What Elements Are Used in Batteries? A Complete Guide to Battery Components
Batteries are an essential part of modern life, powering everything from smartphones and laptops to electric vehicles and renewable energy systems. But have you ever wondered: what elements are in batteries? Or which elements are used in batteries? In this guide, we'll explore the elements used in batteries, from their construction to their role in the efficiency and longevity of various battery types.
What is Inside a Battery? Understanding Battery Components
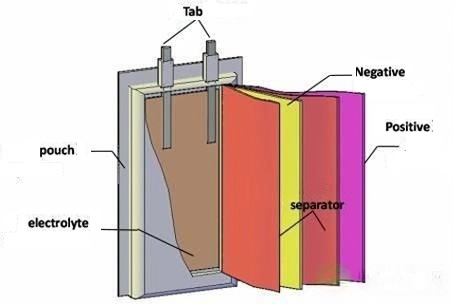
Batteries are made up of several key elements that work together to store and release energy. These elements include the anode, cathode, electrolyte, and separator, each playing a critical role in battery performance.
Anode: The anode is responsible for storing energy and allowing electrons to flow through the circuit. It is typically made from metal or carbon-based materials. Common battery elements for the anode include graphite, silicon, and lithium metal.
Cathode: The cathode releases electrons when the battery discharges, helping power the device. Battery elements like lithium cobalt oxide and lithium iron phosphate are commonly used in cathodes.
Electrolyte: This material allows ions to move between the anode and cathode during charging and discharging, facilitating the battery's energy flow.
Separator: Prevents direct contact between the anode and cathode, which could cause a short circuit, while allowing ions to pass through.
What Element is Used in Batteries? Common Battery Materials
Several elements are commonly used across different types of batteries, each chosen for their ability to enhance the battery's efficiency, energy density, and lifespan. Let’s look at the most widely used elements in batteries.
1. Lithium-Based Elements in Batteries
Lithium Cobalt Oxide (LiCoO₂): Found in lithium-ion batteries, it is known for its high energy density and voltage. It is a popular choice for smartphones and laptops.
Lithium Iron Phosphate (LiFePO₄): Common in high-power batteries, this element is known for its safety and long cycle life.
Lithium Manganate (LiMn₂O₄): Offers a safer, more cost-effective option for low-power devices but comes with limitations in capacity and temperature performance.
Lithium Titanate (Li₄Ti₅O₁₂): Used in high-power applications, known for its long life and safety, but with lower capacity.
2. Nickel and Cobalt Elements in Batteries
Nickel-Cobalt-Aluminum (NCA): Used in electric vehicle batteries, this material provides high energy density but is more expensive.
Nickel-Manganese-Cobalt (NMC): Also used in electric vehicle batteries, NMC offers high energy density and is an alternative to NCA for longer cycle life.
3. Carbon and Graphite Elements in Batteries
Graphite: Most commonly used in lithium-ion batteries for the anode material, graphite is stable, affordable, and offers good cycling performance.
Silicon-Based Anodes: Silicon is gaining popularity due to its high energy capacity, though it suffers from volume expansion during charging.
4. Metal Elements Used in Rechargeable Batteries
Nickel (Ni): A key metal element used in rechargeable batteries like Nickel-Cadmium (NiCd) and Nickel-Metal Hydride (NiMH). Nickel is chosen for its ability to improve charge retention and capacity.
Cadmium (Cd): Found in NiCd batteries, offering durability but suffering from memory effect and lower capacity.
Zinc (Zn): Used in zinc-carbon and alkaline batteries, zinc is a commonly used anode material due to its availability and cost-effectiveness.
5. Lead Elements in Batteries
Lead (Pb): Used in lead-acid batteries, commonly found in vehicles, backup power systems, and uninterruptible power supplies (UPS).
6. Manganese and Other Elements in Batteries
Manganese (Mn): Used in batteries such as lithium-manganese batteries, manganese increases battery stability and longevity.
What Elements Are Used in Different Battery Types?
Lithium-Ion Batteries
Elements Used: Typically use lithium-based materials like lithium cobalt oxide (LiCoO₂) or lithium iron phosphate (LiFePO₄) for the cathode and graphite for the anode.
Applications: Powering smartphones, laptops, electric vehicles, and energy storage systems.
Nickel-Cadmium (NiCd) Batteries
Elements Used: Nickel oxyhydroxide (NiOOH) in the positive electrode and cadmium (Cd) in the negative electrode.
Applications: Power tools, emergency lights, older mobile devices.
Lead-Acid Batteries
Elements Used: Lead dioxide (PbO₂) for the cathode, sponge lead (Pb) for the anode, and sulfuric acid (H₂SO₄) as the electrolyte.
Applications: Used widely in vehicles, backup power systems, and large-scale power storage.
Zinc-Carbon Batteries
Elements Used: Zinc (Zn) for the anode and manganese dioxide (MnO₂) for the cathode.
Applications: Common in flashlights, remote controls, and clocks.
Nickel-Metal Hydride (NiMH) Batteries
Elements Used: Nickel oxyhydroxide (NiOOH) for the cathode and a hydrogen-absorbing alloy for the anode.
Applications: Used in digital cameras, hybrid vehicles, and other portable electronics.
Alkaline Batteries
Elements Used: Zinc (Zn) for the anode and manganese dioxide (MnO₂) for the cathode.
Applications: Powering toys, remote controls, and small electronic devices.
Conclusion: What Elements Are Used in Batteries?
The elements used in batteries depend on the type of battery and its intended application. From the lithium in lithium-ion batteries to the lead and sulfur in lead-acid batteries, the battery elements used determine the battery’s capacity, lifespan, charging speed, and safety.
As battery technology advances, new metal elements used in rechargeable batteries are continuously being explored to make devices more energy-efficient, environmentally friendly, and cost-effective. Whether it's improving the metal elements used in rechargeable batteries or exploring alternatives like silicon-based anodes, the future of battery technology looks promising.

